Last year, in the absence of a cure or vaccines for Coronavirus infection, plasma therapy was administered as a potential treatment for critically ill patients. Seemingly safe and approved by the Food and Drug Administration (FDA) of United States Plasma therapy turned into the most prevalent method to treat critical Covid-19 patients. On Monday, it was dropped from treatment guidelines for Covid-19. With all the uproar surrounding plasma therapy, it is only natural to assume that one might be curious about the said therapy. Here's everything you need to know about Plasma therapy.
What Is Plasma Therapy?
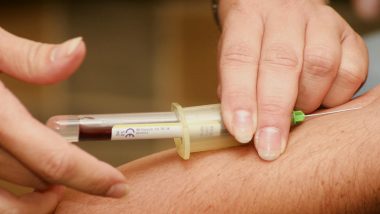
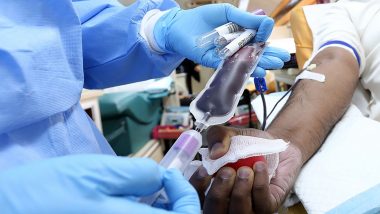
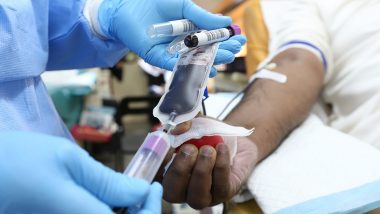
Plasma Therapy - also known as Convalescent Plasma Therapy - involved the transfusion of antibodies from someone who had recovered from COVID-19. It was widely believed patients who had recovered from the disease will have developed antibodies that helped them fight the disease. These antibodies can be extracted by a recovered Covid-19 patient and administered to a critically ill patient to help neutralise the virus.
In order to extract the plasma, blood is drawn from the donor ( person who has recovered from Covid-19) to check for the presence of antibodies. Once checked, the individual can donate plasma. The blood is first extracted and subsequently, plasma is separated from the blood. After the plasma is separated from the blood, the blood is infused back into the donor and only the plasma - now rich in antibodies - is administered to the patient.
Did It Work?
Last year, a study titled ‘Convalescent Plasma in the management of moderate COVID-19 in adults in India,' conducted by ICMR, involving 464 adults said that Plasma Therapy had limited effect in combating the disease or slowing down its progression in their patients. However, despite evidence to the contrary, Plasma Therapy was still prescribed to treat the patients. Over several months, people found their social media handles inundated with desperate pleas for Plasma donors from doctors, caregivers, families of critically ill Covid patients.
Why was it axed?
A recent study in the British medical journal, The Lancet reported, published on May 14th showed in a trial that involved about 5,000 patients from the United Kingdom exhibited no evidence of recovery from Plasma Therapy.
On Monday, the Indian Council of Medical Research (ICMR), dropped the use of Plasma Therapy from the recommended treatment guidelines for COVID-19. The decision comes three days after The Lancet study was published. The researchers in the study said “In patients hospitalised with Covid-19, high-titre convalescent plasma did not improve survival or other prespecified clinical outcomes,”
The study further revealed that they saw no benefit of administering plasma therapy in any patient.
(The above story first appeared on LatestLY on May 18, 2021 03:31 PM IST. For more news and updates on politics, world, sports, entertainment and lifestyle, log on to our website latestly.com).













 Quickly
Quickly